Life sciences organizations are moving quickly from AI experimentation toward embedded AI — but getting there is not simple.
Most companies are no longer asking whether AI matters. They are asking harder questions: Which use cases are worth funding? How do we govern AI responsibly? How do we protect IP, patient data, and regulated content? How do we defend AI-enabled processes when an inspector walks through the door?
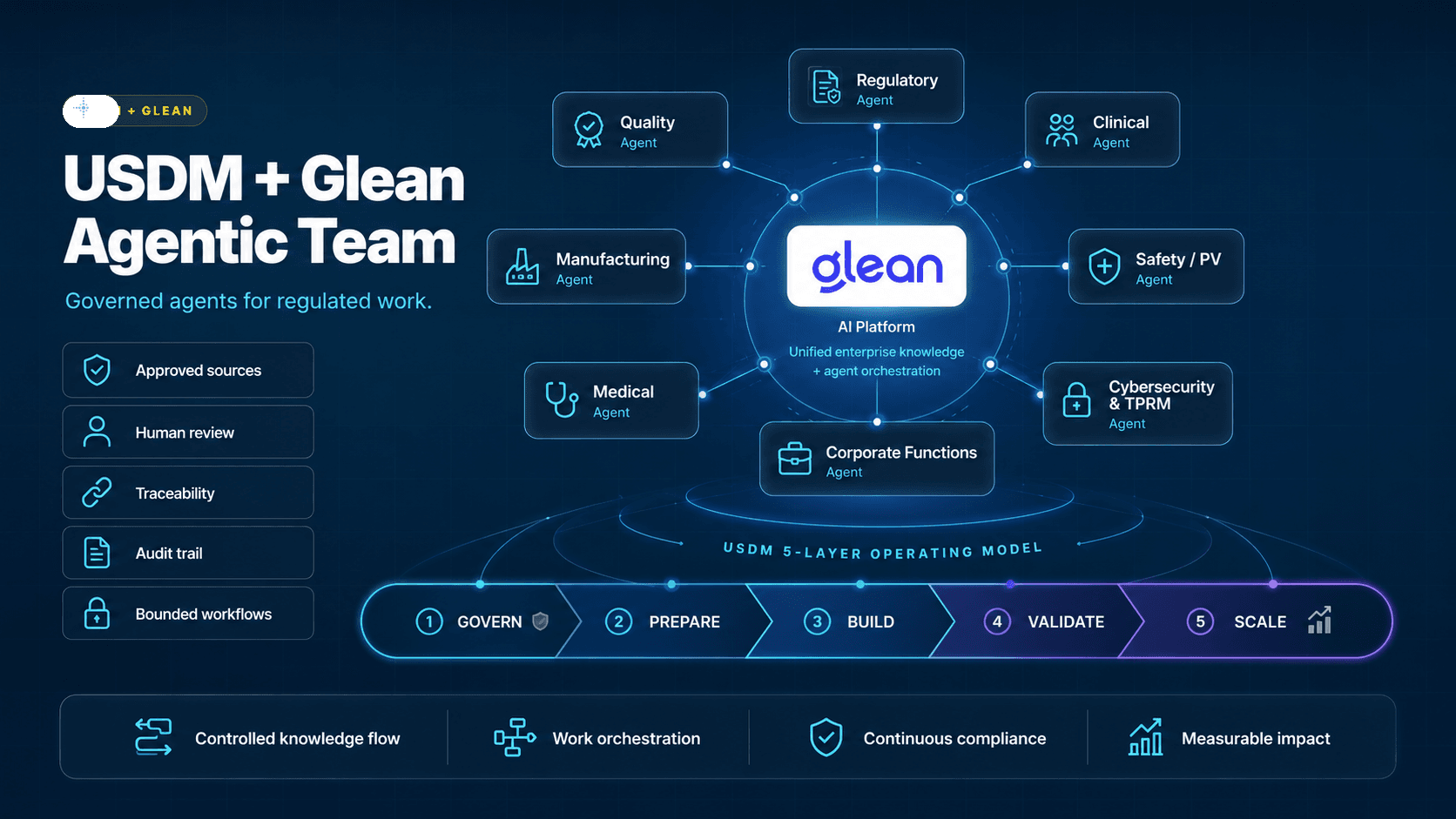
At USDM Life Sciences, we believe the next phase of enterprise AI will depend on trusted knowledge access, strong governance, and practical adoption. That is why we are excited about our partnership with Glean.
Glean's Work AI platform brings enterprise search, AI answers, agents, and connectors into a single experience. USDM brings 25 years of life sciences experience across GxP compliance, validation, quality, regulatory, clinical, manufacturing, and digital transformation.
Together, we see a practical path for helping regulated organizations move from AI intent to measurable impact.
The Life Sciences AI Challenge: Knowledge Is Everywhere, but Answers Are Hard to Find
That knowledge lives across quality systems, regulatory submissions, SOPs, clinical documents, manufacturing records, safety systems, collaboration platforms, emails, project spaces, and data repositories. In many organizations, it is scattered across dozens of disconnected systems.
The problem is not that teams lack information. The problem is that they spend too much time trying to find, interpret, validate, and act on it.
That creates real business friction:
- Scientists and researchers lose time searching for prior work.
- Quality teams manually compare SOPs, deviations, policies, and inspection findings.
- Regulatory and compliance teams struggle to maintain traceability across systems.
- Executives make decisions without a complete view of enterprise knowledge.
- New employees depend too heavily on tribal knowledge.
- AI pilots stall because the underlying context layer is incomplete.
In regulated environments, this challenge is even sharper. It is not enough for AI to produce a useful answer. The answer must be governed, permission-aware, traceable, and defensible.
Why USDM Loves Glean
USDM evaluates AI platforms through a life sciences lens. We look for more than a strong demo. We look for platforms that can support real regulated work, drive adoption, and fit into the systems our customers already use.
Glean stands out for several reasons.
1. Glean Started with Enterprise Search
Glean's foundation is enterprise search. That matters.
Many AI tools begin with a chatbot experience and then try to solve the knowledge problem later. Glean approaches the problem from the other direction: connect enterprise knowledge first, understand permissions, build context, and then enable AI experiences on top of that trusted foundation.
For life sciences companies, that sequence is important. AI is only as useful as the knowledge it can safely access and correctly interpret.
2. Glean Connects to the Systems Where Work Already Happens
Life sciences organizations do not want another isolated AI tool. They need AI that works across the platforms their teams already use.
Glean supports more than 100 native connectors, including systems such as Veeva, ServiceNow, SharePoint, Salesforce, Slack, Teams, Box, Outlook, Jira, Confluence, and other enterprise applications.
That connector ecosystem is a major advantage because it helps reduce the need to extract flat files, manually prepare data, or build one-off integrations before teams can see value.
3. Glean Respects Existing Permissions
Security and access control are non-negotiable in life sciences.
Glean is designed to respect existing permissions in the source systems it connects to. If a user does not have access to a document in the underlying system, they should not gain access to it through Glean.
That permission-aware model is essential for companies managing confidential IP, patient-related information, quality records, regulatory materials, and commercially sensitive data.
4. Glean Supports Traceability
In regulated environments, traceability is not a nice-to-have. It is the foundation of trust.
Glean can show which documents were referenced, what the user asked, what the AI returned, and how the answer was produced. That kind of interaction history is critical for governance, quality oversight, and audit readiness.
For life sciences leaders asking, “How do I defend this AI use case during an inspection?” traceability is one of the first requirements to address.
In regulated environments, traceability is not a nice-to-have. It is the foundation of trust.
5. Glean Makes AI Easier to Adopt
Adoption is where many AI initiatives fail.
A company can build an impressive agent, but if users need weeks of training before it becomes useful, adoption slows. If using the agent takes as much effort as doing the work manually, people revert to old habits.
Glean's natural language experience is a major strength. Users can ask questions in the language of their work — including acronyms and domain-specific terminology — and receive cited, contextual answers from connected enterprise systems.
That matters because life sciences adoption depends on usability from day one.
Where USDM Adds Value
Glean provides the Work AI platform. USDM helps life sciences companies deploy it effectively in regulated environments.
Our role is to bring the domain context, implementation discipline, and compliance perspective required to turn a powerful platform into business value.
GxP Implementation and Validation
USDM helps organizations implement and validate technology in accordance with GxP expectations, data integrity principles, and relevant regulatory requirements.
For AI-enabled workflows, that includes helping define:
- Use case scope and intended use
- Risk assessment frameworks
- Validation strategy
- Governance and oversight models
- Audit positioning
- Traceability expectations
- Change control considerations
- User access and role design
The goal is not simply to “validate AI” in the abstract. The goal is to make specific AI-enabled workflows explainable, controlled, and defensible.
Life Sciences-Specific Agents and Connectors
Glean supports agents and custom integrations. USDM helps extend those capabilities into life sciences workflows.
Examples include:
- SOP impact analysis
- Quality document review
- Inspection readiness support
- Evidence retrieval
- Risk and gap evaluation
- Deviation investigation assistance
- Quality KPI support
- Regulatory and quality knowledge workflows
Some of these use cases may rely on native connectors. Others may require USDM-built connectors or agents designed for specialized systems and processes.
Adoption and Change Management
USDM helps design programs that drive usage across IT, Quality, Regulatory, Clinical, Commercial, and operational teams. That includes role-based enablement, demo environments, stakeholder alignment, governance education, and support models that help users understand when and how to trust AI-enabled workflows.
Ongoing Managed Services
AI platforms are not “set it and forget it.”
As systems, processes, regulations, and business priorities evolve, organizations need ongoing governance and optimization. USDM supports customers after go-live with managed services, enhancement roadmaps, connector support, agent development, and continued compliance alignment.
Practical Use Case: Inspection Readiness
Inspection readiness is another area where the Glean and USDM partnership has strong potential.
An AI-driven inspection readiness architecture built on Glean can connect systems such as Veeva Vault, SAP, Blue Mountain, health authority correspondence, CRO and CMO quality agreements, stability systems, LIMS, prior inspection documentation, recordings, notes, and other sources.
See concrete AI use cases in our latest white paper, AI in Life Sciences: 47 Use Cases for Quality, Regulatory, Clinical, and Manufacturing Teams.
Inspection Readiness Model
- Evidence retrieval — Agents locate relevant records, prior responses, commitments, SOPs, policies, and supporting documentation across connected systems.
- Knowledge curation — The platform organizes relevant information into a usable inspection support context, reducing manual assembly time.
- Risk and gap evaluation — Teams identify missing evidence, inconsistent language, unresolved actions, or areas requiring SME review before the inspection begins.
- Workflow support — Inspection teams coordinate notifications, SME requests, runner assignments, response drafting, and request tracking from a single interface.
- Governance and traceability — Citations, access controls, audit logs, and source references maintain confidence in how outputs were produced.
The vision: a single interface that helps quality and compliance teams manage inspection preparation and inspection-day response with greater speed, consistency, and control.
Why This Partnership Matters Now
Life sciences companies are past the AI curiosity stage.
Many have already experimented with ChatGPT, Copilot, embedded AI features, point solutions, internal prototypes, or specialized vendor tools. The challenge now is AI sprawl: disconnected tools, inconsistent outputs, redundant spending, uneven adoption, and unclear governance.
Glean helps address that challenge by serving as a unified Work AI layer across enterprise knowledge.
USDM helps make that Work AI layer relevant, compliant, and practical for life sciences.
That combination matters because regulated organizations need more than technology. They need a deployment model that accounts for:
- GxP expectations
- Data governance
- Third-party risk
- Validation
- Audit readiness
- User adoption
- Business process integration
- Role-based access
- Measurable outcomes
The companies that get this right will move faster. The companies that wait for perfect certainty may find that the cost of doing nothing compounds over time.
From AI Intent to AI Impact
The path to enterprise AI maturity typically moves through three stages:
- Experimentation: small pilots, proofs of concept, and early productivity tools
- Point solutions: AI embedded into individual platforms or narrow workflows
- Embedded AI: AI integrated across people, data, systems, and business processes
Most life sciences companies are somewhere between experimentation and point solutions today.
The opportunity with Glean and USDM is to help organizations move toward embedded AI in a responsible, governed way.
That means connecting enterprise knowledge, respecting permissions, designing the right context layer, validating regulated workflows where appropriate, and building agents that support the way life sciences teams actually work.
Ready to Explore Work AI for Life Sciences?
If your organization is evaluating enterprise AI, AI governance, inspection readiness, quality automation, or knowledge management, USDM can help you assess where Glean may fit.
Talk to USDM Life Sciences about building a governed Work AI strategy for your organization.
We can help you identify high-value use cases, evaluate readiness, design a compliant implementation path, and build practical AI workflows that move from intent to impact.
Key Takeaways
- Knowledge-first AI — Glean starts with enterprise search and permissions, then layers AI on top. That sequence matters in regulated industries.
- 100+ connectors — Veeva, ServiceNow, SharePoint, Salesforce, and more. No flat-file extraction required.
- Permission-aware by design — Users only see what they already have access to in source systems.
- USDM adds the regulated lens — GxP validation, governance frameworks, adoption programs, and managed services for ongoing compliance.
- Practical starting point — Inspection readiness is a high-value use case where Glean + USDM can deliver measurable impact quickly.
FAQs About Glean
What is Work AI for life sciences?
Work AI for life sciences refers to AI capabilities that help regulated organizations find, understand, and act on enterprise knowledge across systems such as quality, regulatory, clinical, manufacturing, commercial, and collaboration platforms. The goal is to improve productivity while maintaining governance, security, and traceability.
Why did USDM partner with Glean?
USDM partnered with Glean because Glean combines enterprise search, AI answers, agents, connectors, permissions, and traceability in a platform that aligns well with regulated life sciences needs. USDM adds domain expertise, GxP implementation support, validation strategy, adoption services, and life sciences-specific workflow design.
How does Glean help reduce AI sprawl?
Glean can serve as a unified Work AI layer across enterprise systems, helping companies avoid disconnected AI tools, inconsistent user experiences, and redundant point solutions. Instead of creating isolated AI experiences, organizations can connect knowledge across systems through a governed platform.
Can Glean support GxP-regulated workflows?
Glean can support workflows in regulated environments when implemented with appropriate controls, governance, validation strategy, permissions, and traceability. USDM helps life sciences organizations assess intended use, risk, validation requirements, and compliance expectations for specific GxP use cases.
What are examples of Glean use cases in life sciences?
Potential use cases include SOP search and impact analysis, inspection readiness, quality document review, deviation investigation support, evidence retrieval, regulatory knowledge management, KPI support, onboarding, and enterprise knowledge discovery across connected systems.
Does Glean replace quality, regulatory, or compliance experts?
No. Glean should be viewed as a platform that helps experts find information faster, understand relationships across documents, and support workflows with cited knowledge. Human expertise, review, approval, and accountability remain essential in regulated life sciences processes.